|
8/25/2023 0 Comments Atomic mass of iron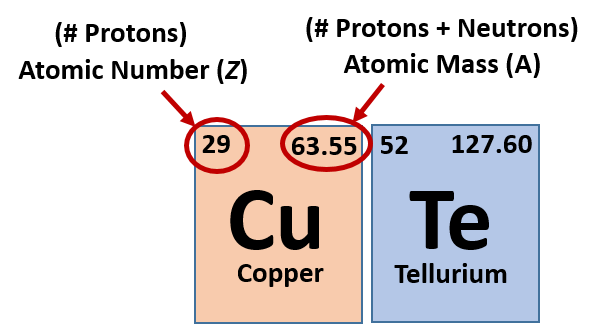
Additional technical, research and safety (MSDS) information is available as is a Reference Calculator for converting relevant units of measurement. Molecular weight calculation: 192.217 + 15.9994 + 14.0067 + (126.90447 + 126.90447 + 126.90447) Percent composition by element Element: Iridium Symbol: Ir Atomic Mass: 192.217 of Atoms: 1 Mass Percent: 31.880 Element: Nitrogen Symbol: N Atomic Mass: 14.0067 of Atoms: 1 Mass Percent: 2.323 Element: Oxygen Symbol: O Atomic Mass: 15. 
Typical and custom packaging is available. American Elements produces to many standard grades when applicable, including Mil Spec (military grade) ACS, Reagent and Technical Grade Food, Agricultural and Pharmaceutical Grade Optical Grade, USP and EP/BP (European Pharmacopoeia/British Pharmacopoeia) and follows applicable ASTM testing standards. Iron Metal 58 isotopic material is generally immediately available. It is by mass the most common element on Earth, forming much of Earth’s outer and inner core. Iron is a metal in the first transition series. For thin film applications it is available as rod, pellets, pieces, granules and sputtering targets and as either an ingot or powder. Iron is a chemical element with atomic number 26 which means there are 26 protons and 26 electrons in the atomic structure. Iron Metal is also available in ultra high purity and as nanoparticles. Iron Metal 58 additionally has special application in the research to develop successful interventions for anemia, conditions for effective iron absorption and excretion, metabolic tracer studies to identify genetic iron control mechanisms, and energy expenditure studies. One formula unit of sodium chloride (NaCl) would weigh 58.44 amu (22.98977 amu for Na + 35.453 amu for Cl), so a mole of sodium chloride would weigh 58.44 grams. The same concept can be extended to ionic compounds and molecules. Iron 58 Metal is one of over 250 stable Metallic isotopes produced by American Elements for biological and biomedical labeling, as target materials and other applications. Thus, since the atomic mass of iron is 55.847 amu, one mole of iron atoms would weigh 55.847 grams. It is both naturally occurring and a produced by fission. We multiply the atomic mass of each isotope by a number representing its relative importance (i.e., its abundance). Iron 58 Metal (Iron-58) is a stable (non-radioactive) isotope of Iron. The average atomic mass of Fe is the weighted average of the atomic masses of its isotopes. Thin Film Deposition & Evaporation Materials.Additive Manufacturing & 3D Printing Materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
